You’re wasting energy right now. You and many of the things all around you.
OK, perhaps it’s unfair to assign such specific agency – it’s not really your fault. Perhaps it’s better to say that energy is being wasted by some vague, abstract agent that shall remain nameless and blameless. Because it just happens.
Your mind may have immediately shifted to thoughts of energy harvesting, conjuring up visions of energy capture from all manner of inadvertent or residual motions: vibrations, steps, waves, wind. But our topic for today, unlike those approaches, involves no motion. Well, no overt motion, anyway. And it doesn’t involve the sun. (At least, not in a direct way.)
As it turns out, differences in temperature represent a source of potential energy that can be harvested. The amounts are small, and the efficiency with which we can extract it is also small (at present, anyway), so it’s not going to replace the internal combustion engine as a source of locomotion. But it can help to make a locomotive – or other heat-generating machine – more efficient by reusing lost heat.
We’re talking about thermal electric generators – TEGs. They’re like solar cells for heat, if you want. Install one somewhere with a temperature difference, and it may reward you with “free” energy.
This topic was spurred by presentations I saw and conversations I had at November’s IDTechEx Energy Harvesting conference. Much of what was discussed might not have seemed relevant to the EE Journal audience, since it was deeply embedded in the world of industrial machines – steel, wiring, exhaust gases, grease. [compulsively washes hands]
But TEGs have significant relevance as these big, raucous Detroit systems blend more closely with delicate Silicon Valley systems. So I figured that a review of how TEGs work, as well as some of the ways they can be – and are being – put to use, would be a productive addition to our energy-harvesting coverage.
TEG Basics
What goes on inside a TEG boils down to what’s called the Seebeck effect: if you take two dissimilar conductors (n- and p-type) and run heat through the junction, the charge carriers diffuse (from the hot to the cold side), and this sets up a voltage. Exactly how many volts you get per degree of temperature difference is captured in the “Seebeck coefficient”; it’s a property of the material, and it may vary with temperature.
Because it’s “better” if you can get more voltage from less heat, good materials for a TEG are ones with high thermal resistance – where a small flux of heat energy creates a big temperature difference – and low electrical resistance – where charge carriers can more easily diffuse to polarize the material and create the voltage. As we’ll see, materials are central to building good TEGs.
There’s actually an inverse effect, too – the Peltier effect – whereby running an electric current through such a junction can cause local heating or cooling. That said, we’ll focus on the Seebeck side of the relationship.
From a practical standpoint, you typically have some source of heat that’s captured in a heat exchanger. A TEG is sandwiched between that heat source and a coolant to maximize the temperature differential (or keep the other side from warming up and neutralizing the difference). The TEG as shown below has several junctions (green/red) stacked together to provide a higher voltage.
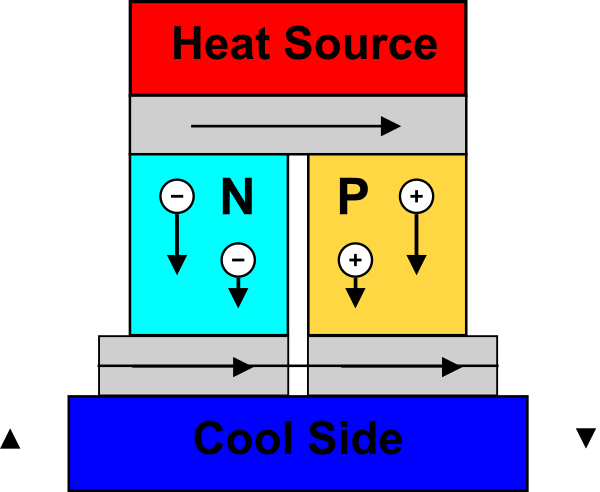
(Image credit: Wikipedia**)
There are a couple of other useful parameters associated with TEGs and their materials. One is the power factor: this is the electrical conductivity multiplied by the square of the Seebeck coefficient. It indicates how much power can be generated, although not necessarily how efficient the generation will be.
Efficiency is indicated by the Greek letter eta (?). It’s simply the energy delivered to a load divided by the heat energy absorbed at the junction. Pretty straightforward.
Less obvious is an overall figure of merit for a material; it’s a semi-intuitive combination of elements with a non-obvious name: ZT*. ZT is a dimensionless ratio: the product of the power factor and temperature divided by the thermal conductivity. So, intuitively, ZT improves with a higher capacity to generate power and a lower ability to conduct heat, but it’s also better at higher temperature (less obvious).
These parameters are the properties of a single material; TEGs are junctions of two materials, with a hot side and a cold side, so there is a modified ZTeff for such junctions, and it’s used in a relation that defines the maximum efficiency possible, ?max. They’re more complicated relations; we don’t need that detail for our discussion.
TEG Materials
We’re deeply embedded in the world of materials science here, something foreign to many of us chipheads. Various different substances have been identified for their utility in TEGs, so we’ll review some of the more popular ones here, summarizing their characteristics.
Bismuth, Lead, and Other Chalcogenides (Tellurides and Selenides)
Bi2Te3 and Bi2Se3 appear to be among the best room-temperature materials, with performance that is independent of temperature. Similarly, PbTe is also used; doping with thallium and selenium improves electrical conductivity.
A superlattice (we saw those recently in our discussion of TRAMs) consisting of alternating layers of Bi2Te3 and Sb2Te3 is claimed to have boosted ZT to 2.4, a very high value, as we’ll see momentarily.
The bismuth-based versions have a relatively limited range of temperatures where they’re effective, and tellurium is one of those rarer (or geographically controlled) materials. Toxicity is high for lead-based compounds, and mechanical strength and thermal stability are also said to be poor.
Skudderites
This gets my award for the least-tech-sounding name I’ve heard in a long time. Almost sounds like it belongs in a children’s song. It’s named after a specific mineral discovered in the Skuterud Mines of Norway, but the working definition has been generalized to include other similar materials.
The composition of a Skudderite is given as LM4X12, where L is a rare earth metal, M is a transition metal, and X is a “metalloid.” A metalloid is a pnictogen. Yeah, that didn’t help me either. Put more simply, it’s an element somewhere between metal and nonmetal, typically in the nitrogen family. Common representatives are phosphorus, antimony, and arsenic.
Skudderites are low in cost and have low thermal conductivity. Based on the number of times I heard them mentioned, I would judge them to be in common use. Their only real downsides appear to be mechanical strength and thermal stability (as compared to other materials).
Half-Heusler Materials
These are alloys said to involve two transition metals and one metal or metalloid (although I’ve seen examples with as many as 5 elements). They appear to be getting a lot of attention as high-temperature generators with reasonable cost and a high power factor.
These materials were described by one company (admittedly, in marketing materials) as performing well with respect to high temperature operation, ease of supply, cost, toxicity, strength, and thermal stability.
I have yet to learn what the “half” signifies, since there are Heusler alloys (defined as specific ferroelectric materials having a particular crystal structure) that seem to be similar to their half-counterparts…
SiGe
This material is the best performer at very high temperatures (around 1000 °C), but it is also relatively expensive, and germanium can apparently be a supply concern.
The following figure illustrates the ZTs available from these materials, just to give you a sense of how they relate (and what typical ZT values are). This is why a ZT of 2.4, as quoted above, would stand out.
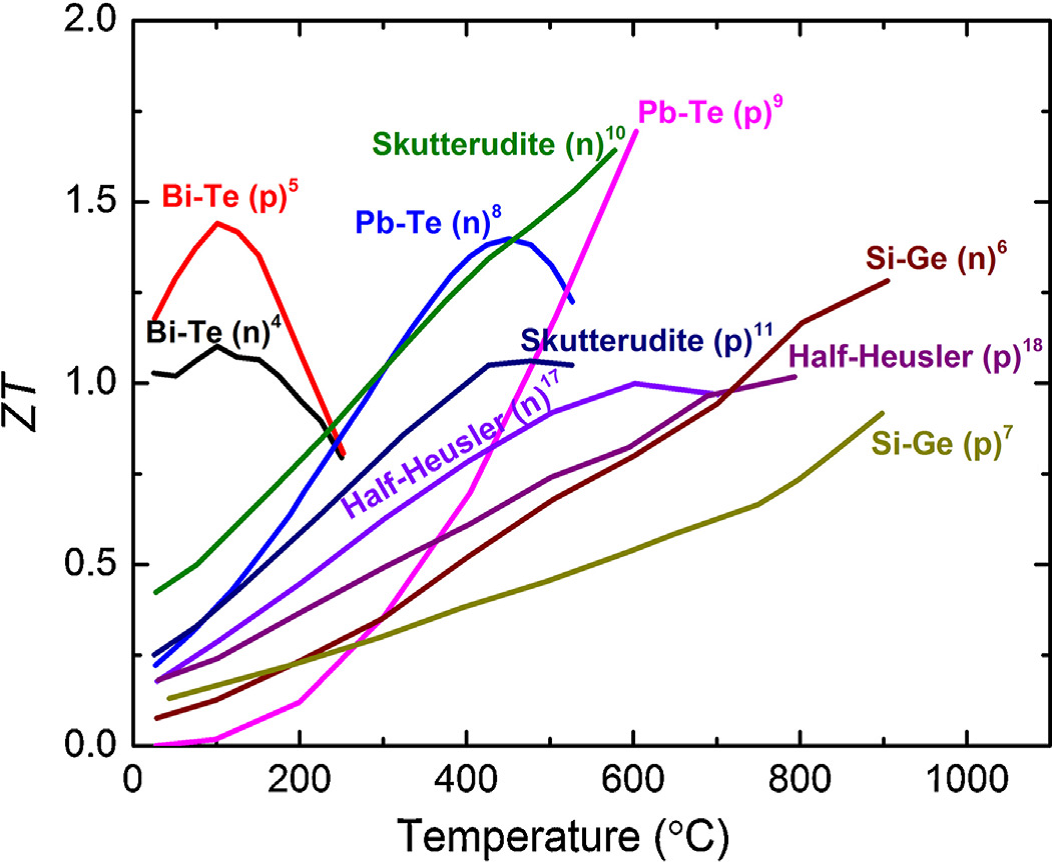
(Image credit Elsevier/Materials Today***)
Meanwhile, in late-breaking news, yet another approach was announced by Sandia National Labs in the last few days. This team is creating arrays of vertical nanowires, and they’re trying to improve the efficiency of past efforts. Their success (so far) has come from a couple of directions. First, and key, was the ability to grow 70-75-nm-wide nanowires that were practically a single crystal with a favorable orientation, grown uniformly and controllably across a large array. The second was the use of an antimony/bismuth alloy to maximize electrical conductivity while minimizing thermal conductivity.
This is still fundamental research; it remains to figure out how best to contact the tops of the nanowires with a high-quality ohmic connection. Presumably we’ll hear more about this if it graduates to commercial use.
Example TEG Systems
Even though TEGs don’t generate a lot of power, they can be useful in running auxiliary equipment or meters or whatnot. In an aggressive industrial setting, running wires can be expensive. Having to string wires is always something of a hassle, but in facilities like refineries, it’s particularly costly: the wires have to be housed in conduit and then be made impervious to gases with gaskets and such so as to be explosion-proof. Running wires undersea or out in the elements creates a similar burden. So being able to generate power locally is particularly useful in these environments.
One specific application I heard about involves integrating a TEG into a boiler. Such an application has obvious heat differentials, and generating it locally means that the electronics needed to run the boiler (including the fan) can be powered by the TEG. Which, of course, means a battery for startup – the chicken to the TEG’s egg (or vice versa).
Why bother? Well, such boilers are often used to heat homes or other buildings. If the power goes out, say, in New England, during an aggressive winter, that can be a serious life-and-death matter. If the boiler is off the grid, however, then, even though the lights may be dark, there is still heat. They’re also putting a USB port onto the system so that people can charge their phones while the grid is down. (Hopefully the cell towers will still be working…)
Vehicles are another area where high heat differences offer up a way to power the increasing number of electronics in the car. According to an IDTechEx session presenter, as much as 40% of auto energy is lost through exhaust gas.
Right now, all the auxiliary components in the vehicle – including electric items powered through the alternator – are ultimately run by clutches and pulleys and gears connected to the crankshaft – that is, they’re powered mechanically by the engine. But the water pump, brakes, power steering, and even the hydraulic pumps in cars and trucks are being shifted to electrical rather than mechanical drive. It appears that scavenging the wasted exhaust heat to power those systems will be necessary for meeting CAFE standards.
Cost is a severe limiter for automotive applications: $1/W was the limit according to one source, a goal similar to that of power plants, which run between $0.60 to $4.00 or so per watt.
One of the IDTechEx presentations described a GM Research project where they took a Suburban and built a TEG system that they could mount underneath along the exhaust pipe. They pushed it up front as much as possible, where the heat was still greatest, but right behind the catalytic converter. (Apparently if they put it before the converter, they run afoul of emissions rules.)
They were able to generate more electricity – 900-1500 W – than the Suburban could use as outfitted – 350 W (calculated via models). What’s the solution? Electrify more of the car – eventually eliminating the alternator if possible (after all, it’s just another load on the engine; the TEG isn’t).
The reason they chose to work on a Suburban, however, reflects one of the challenges of these TEGs: they’re heavy. It’s not so much the materials that are generating the power; it’s the heat exchangers, which tend to involve lots of metal – stainless steel if at very high temperatures. So, at the prototyping level, it took something the size and weight of a Suburban for this to make sense. Getting the overall module smaller and lighter, to the extent possible, will obviously open up more opportunities.
So that’s a quick run-through of the TEG. As with other energy-harvesting approaches, I expect we’ll be seeing more of this over time. We’ll update you as we see progress.
*To date, I haven’t found an explanation of where the name “ZT” comes from.
**”Thermoelectric Generator Diagram” by Ken Brazier – self-made, based on w:Image:ThermoelectricPowerGen.jpg by CM Cullen (which is GFDL 1.2 and CC-by 2.5 licensed). Licensed under GFDL via Wikimedia Commons – http://commons.wikimedia.org/wiki/File:Thermoelectric_Generator_Diagram.svg#mediaviewer/File:Thermoelectric_Generator_Diagram.svg
***Reprinted from Materials Today, Vol. 16, Issue 10, Shuo Chen, Zhifeng Ren, “Recent progress of half-Heusler for moderate temperature thermoelectric applications”, p. 388, Copyright 2013, with permission from Elsevier.





Are you harvesting heat? What approach do you use?
Not harvesting heat but did see one of these:
http://valiantfireside.com/get/premiair-4-blade-heat-powered-fan (other such products are available)
and wondered what material it used. Might just be a bit of a toy. I had assumed it used a thermopile made of non-exotic materials.
Good article, and the third paragraph is right on. Generating power from temperature differences hasn’t been top of mind due to the lack of awareness and finished products. I would like to encourage readers to browse our Perpetua portfolio of thermoelectric energy harvesters that are being used in oil & gas, and other applications. You’ll see how the energy harvesters power wireless sensors with minimal temperature differences.
http://www.perpetuapower.com/products